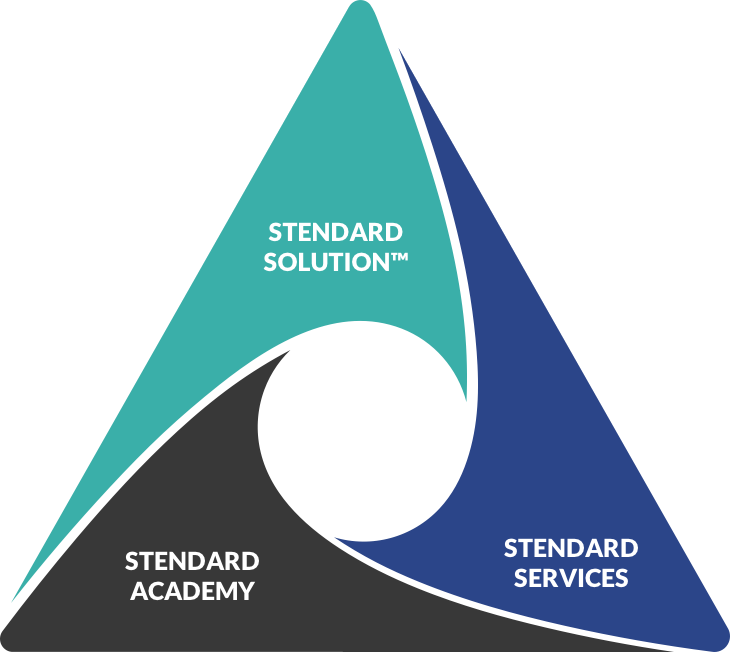
- Consulting
Getting certified with the world's leading food safety management standard.
Getting certified with the global standard used by healthcare industry.
Getting certified with a globally accepted indication of security effectiveness.

Establish a complete and effective medical device regulatory strategy.
Increase product and service quality with Stendard's ISO consulting.
- Software
Summary of our product and features that may help your business process even greater and possibly solve your difficulties in business process.
Use our document generator to generate each Manual, Procedure, Form Templates and etc. Define the step-by-step instructions needed for each operation within the Work Instructions.
A powerful tool that streamlines the monitoring process and helps users make informed decisions based on the data that is most relevant to their needs.
A simple but powerful module for you to store, organise and edit your files seamlessly without any hassle.
With Document Control, never worry about inaccuracy and non-compliance of documentation files and processes ever again.
An effective way to reduce paperwork related hindrance within the workplace and improve organisational efficiency.
With the ability to add, edit, and delete data, import data from external sources, and search and sort data, you can easily organize, analyze, and report on data in a more efficient manner.
Create any form that you need to support your daily activities. As the name suggests, building a form is now a breeze as you drag & drop components in.
Our aim at Stendard is not only to provide you with quality consulting services. We want to empower our clients such as yourself by providing a wide range of ISO related courses.
Create your training plan to group your training courses. By setting up these plan, you can set multiple items to be trained by several team members at once.
Compile and organise essential documents required for audits. With this module, you will be able to breeze through any up and coming audits without fearing missing or incorrect documents!
Our AI engine for Document Classification will help you significantly speed up the document classification process and allow less room for human error when handling these vast amounts of documents.
Changelog will list all the updates and patches we have made to every software update to ensure that you know the new features or updates introduced to the system.
You can ensure that accountability is incorporated into your organisation’s document management system with a robust audit trail system.
A simple but powerful tool to locate every single document you need.
- Academy
- About